BAT shoehorns its nicotine pouches onto the Canadian market
On Tuesday this week, Health Canada made public its decision to issue a natural health food license to a subsidiary of British American Tobacco. As a result, British American Tobacco has become the first manufacturer to be authorized to market nicotine pouches in Canada.
This federal license, issued on July 18, 2023, allows BAT/Nicoventures authority to market five flavours of a 4mg nicotine pouch. It also allows the company to advertise these pouches as a way to quit smoking (“help you quit smoking by delivering nicotine to your body, temporarily relieving cravings and nicotine withdrawal symptoms”) and a way for smokers to cope where smoking is banned (“used when you need to temporarily refrain from smoking, for example, around others, in smoke-free areas, airplanes, or in other situations when you wish to avoid smoking.”) It permits the pouches to be sold to children (although it advises people under 18 not to use it).
This post highlights how this development reveals vulnerabilities in Canada’s health regulatory approach, and how BAT has been able to use regulatory pathways intended for health products as a route to market for its addictive consumer products. Although Health Canada has adopted a harm reduction approach to tobacco, it has failed to established a legal framework for this strategy or to be transparent about how nicotine pouches fit into this strategy. Whatever benefits nicotine pouches might offer for public health, allowing British American Tobacco free rein on advertising them is unlikely to achieve a public benefit.
Summary
Canadian law does not currently allow for the sale of nicotine pouches, other than as regulated medicines. Over the past five years, BAT and other tobacco companies have launched “modern oral” products as a way to expand their range of nicotine products to overcome reduced cigarette use. Their nicotine pouch is sold under the brand name VELO and is currently available in at least 22 countries. BAT is hoping to expand the market for modern oral tobacco beyond the Nordic countries, including in North America.
BAT can use its ownership of NRT companies to gain access to therapeutic channels to sell nicotine. BAT acquired Niconovum, which pioneered nicotine pouches for sale as a form of NRT, branded as ZONNIC. Scandinavian countries have authorized ZONNIC as NRT. However, the product authorization issued by Health Canada shows that the ingredients and flavourings in the Scandinavian ZONNIC are different from the product that BAT will sell in Canada. Based on the ingredient list and flavourings, the product approved by Health Canada this week is almost certainly the 4 mg version of their VELO nicotine pouch.
BAT will face very few restrictions on how it can market ZONNIC nicotine pouches in Canada, other than with respect to how it represents the therapeutic benefits of the product. Under federal law, advertisements will be allowed in all media, and there are no restrictions on the use of lifestyle imagery. Based on historical marketing practices of BAT, we can expect that the product and promotional material will be available in convenience store outlets in most provinces. Provincial governments have the authority to set restrictions on where the product can be sold and how it can be promoted, but few have such measures currently in place. Other companies are positioned to start selling nicotine pouches in Canada, and the approval of BAT’s application may facilitate their doing so. This development raises questions about the adequacy of Canadian law to protect the public from the marketing strategies of tobacco companies, about Canada’s application of Article 5.3 of the Framework Convention on Tobacco Control, and about the need for an integrated nicotine strategy.
Canadian law does not currently allow for the sale of nicotine pouches, other than as regulated medicines.
Three federal regulatory systems are in place which control the sale of nicotine products:
1. By default, nicotine is a prescription drug
Under the Food and Drugs Act, Health Canada establishes which drugs are only available by prescription. Nicotine is currently included on this list, which means nicotine-bearing products can only be sold under prescription unless they are specifically exempted under other laws or regulations. The two current exemptions are tobacco and vaping products (which are exempted in the legislation) and NRT (which is exempted in the Prescription Drug List). The Prescription Drug List is set by Health Canada at an administrative level.
2. NRT is regulated as a Natural Health Product
Certain NRT delivery-systems qualify as natural health products and are regulated under the Natural Health Products Regulations. These include gums, patches, lozenges and NRT in “a form to be administered into the oral cavity by means of a non-active device (one that operates on energy generated by the human body or by gravity).” The maximum allowable nicotine per dose is 4 mg, except for patches which can deliver 22 mg per day. These criteria can be changed by Health Canada.
Manufacturers may not sell NRT products unless they receive authorization from Health Canada. Authorization is given for each product and is contingent on Health Canada being provided with evidence to support safety and efficacy as well as support for health claims. Those products which are authorized are given a Natural Product Number (NPN) which is linked with a specific recommended use and specific risk information and contra-indications. (The NPN for Zonnic pouches is 80125630).
If nicotine were not plant-derived, NRT products would be regulated as non-prescription drug.
3. Nicotine in Tobacco and Vaping Products is regulated as a recreational drug
When nicotine is contained in tobacco or a vaping product, it is regulated under the Tobacco and Vaping Products Act. Products which meet the definitions of this act are exempt from the controls in the Food and Drugs Act, and do not need pre-approval to be marketed. There are many restrictions on how tobacco and vaping products can be promoted and packaged.
Under this set of laws, the only regulatory path open to BAT to legally market nicotine pouches in Canada is as a natural health product. They are, however, restricted to selling products that contain no more than 4 mg of nicotine per dose.
Over the past five years, BAT and other tobacco companies have launched “modern oral” products as a way to expand their range of nicotine products to overcome reduced cigarette use.
BAT and other tobacco companies have responded to the gradual reduction in smoking in ways which seek to increase profitability (so profits stay high while volume sales go down), which seek to expand their range of nicotine products (so they retain customers who would use nicotine but who might not smoke cigarettes). They are also expand their operations into other pharmacological substances (using their expertise with drug delivery systems to build a non-nicotine revenue stream).
Four years ago, BAT identified nicotine pouches as “an exciting new space”, and began to manufacture “modern oral” products by extracting nicotine from tobacco plants and then blending it with bleached tree fiber (cellulose) and flavourings. In addition to giving the impression of being less risky, these nicotine products had the advantage of not being subject to tobacco taxes. (Taxes on a tin of oral tobacco in Canada are about $20).

BAT’s nicotine pouch is sold under the brand name VELO and is currently available in at least 22 countries.
Originally called EPOK, LYFT and VELO, BAT has now branded its nicotine pouches under the VELO trademark.
Over the past 4 years, BAT has expanded its marketing of VELO nicotine pouches to 22 countries (17 of which are in Europe). Typically, these are sold in a dozen or more flavours, and in strengths ranging from 4 mg to 17 mg of nicotine per pouch.
BAT is hoping to expand the market for modern oral tobacco beyond the Nordic countries, including in North America.
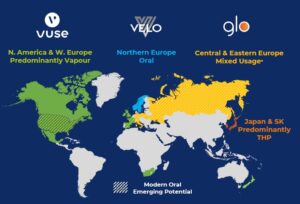
BAT initially focused different alternative-nicotine products in different parts of the world: In Europe and North America, its emphasis has mostly been on vaping products. In the Nordic countries, where snus use was established, it has focused on modern oral. In Japan it has focused on heated tobacco products (its i-glo heated tobacco product was sold for a brief time in Canada). In Central and Eastern Europe the company has been equally focused on heated tobacco and vaping market. (BAT, slide 33)
This year it has been reported that in response to additional restrictions on vaping products, BAT and other tobacco companies have increased efforts to include nicotine pouches in their product offerings. Even in countries without an oral tobacco tradition (such as the United Kingdom and Europe), BAT is promoting VELO using lifestyle imagery and messages about as an alternative reduced-risk product.
BAT and other tobacco companies have bought out independent companies which made nicotine replacement therapies.
Tobacco investments in NRT products include BAT’s 2010 acquisition of marketing rights for VOKE (a nicotine inhaler), Philip Morris International’s 2021 purchase of Fertin Pharma, and the 2023 acquisition by Imperial Brands of the start-up Canadian nicotine-pouch manufacturer TJP .
Preceding these purchases was the 2009 purchase by Reynolds American (RAI) of the Swedish company Niconovum, which developed the ZONNIC brand nicotine pouches and other NRTs. For a time, RAI sold ZONNIC-brand gum in the United States through convenience channels, and was the first tobacco company to be authorized by the FDA to sell stop-smoking medication. When BAT purchased Reynolds American in 2017, it also acquired Niconovum. In 2019, ZONNIC gum was withdrawn from the U.S. market.
Although BAT owns Niconovum, it is using a different subsidiary, Nicoventures, to sell ZONNIC in Canada. Nicoventures is the branch of BAT that is responsible for selling VUSE vaping products. Nicotine pouches will not be sold in Canada by a pharmaceutical branch of a multinational conglomerate, but by a company that has a mission to increase nicotine use.
Scandinavian countries have permitted the use of mint-flavoured 4 mg nicotine pouches as a form of NR, and BAT acquired the company which pioneered that form of NRT.
Although Many governments permit the sale of nicotine pouches, in only a few are they sold with medical claims or as a form of NRT.
Niconovum reports that Zonnic pouches are currently sold as NRT in the Nordic countries of Sweden, Norway, Finland and Iceland. In these countries it is available in only one flavour (Mint)
The pouch was approved for sale by the Swedish drug authority in 2012 , and an assessment record for that decision is available. The package leaflets which are available from medicine authorities in Sweden, Norway, Finland and Iceland contain information on the regulator’s views of the risks and uses of the product, and also the ingredients used.
In Canada, BAT is using the brand name ZONNIC of the Scandinavian-approved pouch NRT but has received Health Canada authority to market a product that has different ingredients and flavourings.
The ingredient lists on the Scandinavian package leaflets and the Health Canada authorization show that the ZONNIC product licensed for sale in Canada is not the same as the one licensed for sale in Nordic Countries. Instead, BAT’s Canadian natural health product nicotine pouches are made with the same ingredients that they use for the VELO recreational nicotine they sell in other countries.
The following ingredients are listed for ZONNIC in the package leaflet authorized in Sweden and other Scandinavian countries: Active substance: nicotine – 4 mg; Other ingredients: cellulose (microcrystalline), ascorbyl palmitate (E304), trisodium phosphate, acesulfame potassium (E950), aspartame (E951); Flavourings: mint flavour
The following ingredients for ZONNIC are identified in Health Canada’s natural health product database: Medicinal ingredient: 1-Methyl-2-(3-pyridyl)pyrrolidine [nicotine] – 4 mg; Non-medicinal ingredient: Microcrystalline cellulose; Propylene glycol; Sodium alginate; Sodium bicarbonate; Sodium chloride; Sucralose; Water; Xylitol; Flavourings: Berry flavour; Cranberry flavour; Mint flavour; Spearmint flavour, Topical citrus flavour
An ingredient list is provided for VELO pouches on some retail websites (although BAT’s information is less specific). Nicotine; Microcrystalline cellulose fibers (E460 (i)), propylene glycol (E1520), sodium alginate (E401); sodium bicarbonate (E500), salt (sodium chloride), sucralose (E955), water, Xylitol (E967); Flavors

Based on the ingredient list and flavourings, the product approved by Health Canada this week is almost certainly a 4mg version of their VELO product.
Health Canada has authorized BAT/Nicoventures to sell Zonnic in the following five flavours: Polar Mint; Berry Frost; Chill Mint; Cranberry Fizz; Tropic Breeze.
Some of these flavours – tropic breeze, polar mint – are identical to those sold with VELO in other markets.
There are currently very few restrictions on how BAT can market these nicotine pouches in Canada, other than with respect to how it represents the therapeutic benefits of the product.
In Canada, federal law puts severe limits on most direct-to-consumer advertising of prescription drugs, as it does for tobacco and vaping products. There are fewer restrictions on advertising for non-prescription or health products.
As a result, advertisements for BAT’s nicotine pouches will be allowed on television, in video-games, in corner stores, on social media and in other places where young people have access.
Health Canada has approved Guidelines for Consumer Advertising of Health Products. These explain that the constraints that BAT will face in promoting ZONNIC pouches mostly relate to ensuring that the advertising claims are consistent with the license or authorized labelling.
Although the authorization counsels consumers to not use the product if they are under 18 there are no age restrictions on the purchase of natural health products.
Unless there are legal changes, BAT will be allowed to advertise nicotine pouches in all media, with no restrictions on the use of lifestyle imagery, contests, give-aways or other promotions.
BAT has the economic power to distribute and promote nicotine pouches through the 29,000 retail outlets with which they currently have distribution contracts.
Provincial governments have the authority to set restrictions on where the product can be sold and how it can be promoted, but few have such measures currently in place.
Provincial governments have coordinated their regulations on non-prescription pharmaceutical products, but do not have a coordinated system for natural health products. They do retain authority over retailing, and can impose restrictions on if, how and where nicotine pouches can be sold or promoted.
For example, Quebec regulations do not allow the sale of conventional NRT except in pharmacies, but the wording of the regulation did not anticipate nicotine pouches, and refers only to “chewing gums, inhalers, lozenges or transdermal nicotine replacement patches.”
Other companies are positioned to start selling nicotine pouches in Canada, and the approval of BAT’s application may facilitate their doing so.
According to Health Canada officials, more than two dozen applications have been received for market authorization for nicotine pouches, some of which have been refused and some of which are still under review.
Those applications which were refused to date failed to provide sufficient evidence that the products were effective as stop smoking aids. Now that the department has accepted the evidence provided by BAT/Nicoventures, the road for other applications will be smoother.
Health Canada faces limits in its ability to refuse authorization: companies can (and do) successfully challenge refusals to grant authorization to natural health products. One of these contested decisions has involved a stop-smoking aid (Resolve).
This development raises questions about the adequacy of Canadian law to protect the public from new nicotine products.
Health Canada has indicated that the results of a clinical trial were submitted with BAT/Nicoventure’s application, but further information is not known at this time.
There has been one published Randomized Clinical Trial of Zonnic as a stop-smoking aid, which was conducted in New Zealand and published in Nicotine and Tobacco Research over 14 years ago. The study was paid for by Niconovum,
This study provides a very slender evidence base on which to permit a new category of nicotine products on the market. It involved only 28 people who agreed to abstain from smoking for three days on each of three occasions: one time using a placebo pouch, another using NRT gum and a third time using Zonnic nicotine pouches. Twenty-one of 28 people abstained from smoking for 3 days using 4 mg Zonnic – a better result than was obtained with 4 mg NRT gum.
There are no other published clinical trials of ZONNIC or other non-tobacco nicotine pouches as smoking cessation aids currently identified on Pubmed, although there are studies which compare the way the body receives nicotine from pouches with other NRT. The inventor of Zonnic (Karl Fagerstrom) promotes pouches (NP) as a harm reduction product, but recently acknowledged that “The NP have so far not been well researched and there are only a few papers published.”
This decision raises questions about Canada’s application of Article 5.3 of the Framework Convention on Tobacco Control .
Like the recent Medicago debacle, Health Canada is again caught in the position of licensing medical products manufactured by tobacco companies. As these companies expand into medically-regulated products, so does the potential for governments to breach their obligations to protect public health from tobacco industry interference. This is especially relevant with respect to stop-smoking medications, which could permit tobacco companies to engage in public relations efforts to forestall tobacco regulations.
This development is a reminder that Canada needs an integrated nicotine strategy.
As with SESH+ nicotine gum, this stop-smoking product is a sharp reminder that Canada needs an integrated nicotine policy. Health Canada has not yet acknowledged the need for such reforms.









