Health Canada’s vaping flavour ban is still missing in action.
It’s time to admit it is dead.
 In June 2021 Health Canada stated its intention to reduce youth vaping by removing flavourings from vaping liquids, and published draft regulations to that effect.
In June 2021 Health Canada stated its intention to reduce youth vaping by removing flavourings from vaping liquids, and published draft regulations to that effect.
Predictably, the release of the draft regulations was followed with noisy and prolonged protests and intensive lobbying by those in the business of selling these liquids. More unexpected perhaps was the radio silence and prolonged inaction on the part of government following that initial release.
More than 600 days after federal officials last gave any sign that they intend to finalize these regulations, it now seems prudent to conclude that the flavour ban has been left to die on the vine.
This post reviews events that support this conclusion, and contrasts the lack of movement in Canada with measures underway in other countries.
Background: When Ottawa decides to say “no”, the record of that decision is sealed.
It is rare for Ottawa’s decision-makers to be transparent about their decisions to turn down policy recommendations. Outside of Parliamentary votes, the system of federal policy-setting is set up to make public those decisions which make regulatory changes, and to withhold the existence of decisions to reject such proposals.
This secrecy is enshrined in law: the Access to Information Act (s. 21) prevents disclosure of unsuccessful departmental recommendations for 20 years, and the existence of Cabinet decisions to reject regulations are secret until they are declassified 20 years later. (Decisions that result in change are made public through Orders-in-Council and publication in the Canada Gazette).
With these constraints in place, it is impossible to state for certain that Health Canada, the Minister or Associate Minister of Health, the Privy Council Office, the Prime Minister’s office or the federal Cabinet has stalled or rejected the idea of protecting the public from the sale of flavoured vaping liquids.
When Ottawa says “no”, it is expressed as “not yes” or “not yet”.
In the absence of records of decisions on the vaping flavour issue, it is necessary to interpret the actions – and absence of actions – that are on the public record. These include how officials refer to the regulatory proposal in mandatory reports, and how they fail to refer to it in proactive voluntary communications.
Since the fall of 2021:
* Finalizing the flavour regulations was conspicuously missing from the mandate letters issued by the Prime Minister to the Minister of Health and the Associate Minister in December 2021.
* Among the thousands of tweets issued by Health Canada, the Minister of Health and the Associate Minister of Health, there is only one mention of youth vaping, and no mention of the need to protect young people from vaping flavours.
* Departmental communications have not acknowledged the role that flavours play in inducing young people to vape, but have been limited to references of the department “reviewing feedback”.
* In the department’s Legislative Review of the Tobacco and Vaping Products Act, tabled in Parliament last December, the department prioritizes its areas for potential action, but makes no commitment to finalizing flavour restrictions. Instead, the report demurred that “More time is needed to fully assess the impact of these recent regulatory initiatives.”
* Although Departmental Plans identified “finalizing” flavour restrictions in 2021-2022, plans were downgraded to “ongoing work on the proposed order” in 2022-2023. The 2023-2024 Departmental Plan makes no reference to the idea of restricting flavours.
* In April 2022, the department’s Forward Regulatory Plan was modified and no timeline was given for the completion of this regulation, although timelines were provided for other tobacco and vaping regulation.
The importance of banning flavours has not changed.
The rationale provided by Health Canada for banning flavours in June 2021 is no less compelling today.
* The government continues to have a statutory responsibility to protect young persons and non-users of tobacco products from inducements to use vaping products and from exposure to and dependence on nicotine.
* Flavours continue to induce young people to try vaping and to continue vaping. Even researchers who are supportive of relaxed regulatory controls on e-cigarettes acknowledge that young people who first use a flavoured e-liquid product are more likely to continue vaping.
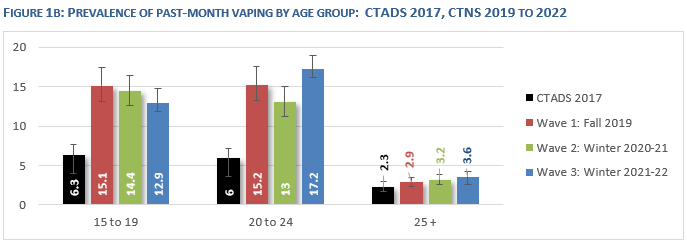
* Statistics Canada reports that the prevalence of vaping among teenagers has not gone down (and it has increased among young adults). There are still over half a million Canadians who have never smoked but who are vaping.
* As reported here earlier, the marketing of flavoured vaping products is increasing.
Meanwhile, other governments have said “yes” to protecting youth from vaping flavours
In the 18 months that Health Canada has been “reviewing feedback” and “assess[ing] the impact” of measures to stop flavours from inducing children to try vaping, a number of other countries have adopted flavour restrictions. Restrictions are already in place in the Netherlands (January 2023), China (October 2022), Lithuania (July 2022), Denmark (April 2022), Hungary (May 2020), Estonia (July 2019) and Finland (2016). (See downloadable fact sheet). Although mired in legal challenges, the U.S. Food and Drug Administration has consistently ruled against the marketing of flavoured e-cigarettes, saying that their appeal to youth makes doing so inappropriate for the protection of public health.
In addition to those countries which only allow tobacco-flavoured liquid, tehre are 30 or more countries which ban the sale of all e-cigarettes, including unflavoured or tobacco-flavoured liquids. Among these are the large nations of Mexico, Brazil, India, Turkey and Japan.

All leading Canadian health organizations involved in tobacco control have called for an end to the sale of flavoured vaping liquids in the consumer goods market (i.e. vaping products which are neither authorized nor sold as smoking cessation products). Among many others, these include the Canadian Cancer Society, the Canadian Council of Chief Medical Officers of Health (CCMOH), the Canadian Dental Hygienists Association, the Canadian Lung Association/Canadian Thoracic Society, the Canadian Medical Association (CMA), the Canadian Paediatric Society (CPS), and Heart and Stoke.
The policy is also supported by international health bodies, including the major American and European Health Charities. The World Health Organization study group on tobacco product regulation has called for the flavour restrictions that are in place for tobacco to be extended to all nicotine products. (A sample of positions can be downloaded here).
Regulatory restrictions in other countries are helping protect youth. A just-published review of youth vaping in 75 countries that participate in the standardized Global Youth Tobacco Survey (which does not include Canada) found that in comparison with youth in countries without regulatory policies “youth in countries with the most restrictive e-cigarette regulations … had 0.6 times lower odds of being current e-cigarette users (aOR 0.6, 95% CI 0.6, 0.7)”.
It is possible that the decision against flavour restrictions was made because federal officials believe both that e-cigarettes help smokers quit at the population level and also that flavourings are necessary for such a success.
No evidence has been offered by government to support this conclusion, other than a reference to a review of randomized clinical trials which excluded the results of population level studies. Such a position would be at odds with scientific reviews by many other health authorities, including the World Health Organization, the U.S. Surgeon General, and the College of Family Physicians of Canada. It would also appear to pre-empt a decision of the Canadian Task Force on Preventive Health Care which is expected later this year.
If the desire to provide smokers with access to flavoured liquids is the reason Health Canada has backtracked on its flavouring regulations, they could and should find other ways to protect young people from accessing these products. One obvious option is to restrict the supply of vaping products to the therapeutic channels in which there is oversight by health professionals. This could be done by requiring a prescription, as Australia does, or by selling vaping liquids without prescription in from ‘behind the counter’ in pharmacies.
The apparent demise of the flavour ban is a set-back, made worse by the lack of transparency about the reasons behind this policy reversal and the absence of discussion on alternative ways to protect youth.



